

Regulatory movement around PFAS is picking up; this year and next could be monumental around managing these toxic compounds in landfills and leachate. Operators should look out for proposed U.S. Environmental Protection Agency (EPA) rules in 2022 and final rules in 2023. Most notably, two PFAS categories, PFOA and PFOS, could be classified as hazardous wastes under the Resource Conservation and Recovery Act (RCRA) and the Comprehensive Environmental Response, Compensation, and Liability Act (CERCLA), aka Superfund. Also, expect rules on monitoring and limiting PFAS in drinking water.
Amidst this regulatory activity, PFAS treatment research advances, which will be critical to landfill operators when they are charged with managing this very challenging stream. With existing options, it’s near impossible to destroy these “forever chemicals,” known for their carbon-fluorine bond, considered one of the strongest in nature.
SCS Engineers’ Gomathy Radhakrishna Iyer advises operators on what to look for to brace for regulatory change and advises them on their best defense—the treatment piece. She explains current options and potential technology breakthroughs on the horizon.
“On the legislative front, standardized guidance might not happen overnight. There’s much to learn, as leachate is not the same, including as it pertains to PFAS. Concentrations and compounds vary. So, EPA is gathering data and knowledge to inform policy and mitigation options moving forward,” Iyer says.
Today’s focus entails developing and validating methods to detect and measure PFAS in the environment. The EPA is evaluating technologies to reduce it and is trying to understand better the fate and transport of PFAS in landfills (including landfill gas, leachate, and waste).
While PFAS concentrations in leachate sent to publicly owned treatment plants (POTW) are unknown, the EPA 2023 rule aims to fill in the missing pieces. What is learned and subsequent decisions will be critical to landfill operators who depend on POTWs as a final destination for leachate and at a time when POTWs meet stringent guidelines on what they can accept. The EPA’s focus will begin with guidance on monitoring and reporting figures, including a list of PFAS to watch for in 2022.
In the meantime, the agency published interim guidance on destroying and disposing of PFAS, which it plans to update in fall 2023. The interim guidance identifies the information gap with regard to PFAS testing and monitoring, reiterating the need for further research to address the FY20 National Defense Authorization Act NDAA requirements. Operators can also look to SWANA treatment guidelines to help prepare for new rules.
Get ahead of the game by doing your homework on treatments, Iyer advises. POTWs have discharge limits, and once PFAS in leachate is weighed in with the existing constituent limits on permits, ensuring a disposal destination will call for proactive measures.
The discussion on treatments will be important. Iyer advises on staying up with expectations that may be in the pipeline, beginning by focusing on today’s commercially available options:
Comparing these methods, Iyer says, “Biological treatments work better simply as a pretreatment method, removing PFAS to some extent. Their performance may also only apply to non-biodegradable organic matter. Considering these limitations, the alternative of physical-chemical treatments is most often recommended by industry experts; they appear to be more effective as supported by data,” Iyer says.
Her preference is RO, the membrane-enabled separation process, which many treatment plants already use, or are considering, to remediate other constituents. “Because we know RO to be effective with other contaminants and PFAS, I think it’s a great gainer, especially if plants already use this method to treat leachate for other contaminants successfully,” she says.
RO requires relatively little operational expertise, while other physical-chemical methods, such as GAC and ion exchange, require some chemistry knowledge.
“With granular activated carbon and ion exchange, resins attach to contaminants in leachate. These approaches require pretreatment for organics removal, process understanding, and operator involvement. Conversely, with RO, you learn a fairly straightforward process and move through the steps,” she says.
But while physical-chemical treatments are the best readily available options today, each has limitations. RO leaves a residue requiring further treatment; then, the material is typically recirculated in landfills as a slurry or hauled to a POTW, meaning there is no guarantee they will not need to be addressed later. Other methods, such as GAC, are more energy-intensive and have limited sorbent capacity. Ion exchange, in particular, has difficulty removing short-chain PFAS, which persist in the environment.
When the time comes that PFAS have stringent discharge limit requirements, no one of these technologies may work as a standalone, so the search is on for more robust systems.
Several new treatments are under research; unlike their predecessors, they appear to break the chemical bond.
Iyer shares her take on each option:
“I’m especially interested in seeing how plasma treatment works in the real world versus the lab. The building costs can be higher, and leveraging electricity to break the bond is expensive. But the maintenance should be easy and relatively inexpensive compared to other technologies. It will be interesting to see how economical it would be for landfills over the long run.”
There is more to learn about each of these new technologies. Researchers are working to identify the adsorbents that best suit PFAS compound removal, whether short or long chains. With photocatalytic reaction, a research direction is exploring combining UV rays, a catalyst, and an oxidant to degrade PFAS.
“We know that the absorption options and photocatalytic concepts work well on strong contaminants,” Iyer says. She moves on to her thoughts on thermal treatment. She wants to know more about this particular option before weighing in. “I’m not sure how feasible this method will be for the operators. PFAS get destroyed at a temperature greater than 1,000 degrees Celsius. But for high quantities of leachate, this option could be expensive.”
Most EPA-funded research is based on these developing treatment processes. But there is plenty to evaluate to identify the best solutions in a given scenario. With that understanding, the agency is trying to understand the types and volumes of PFAS generated, how they change or degrade as they enter landfills, and where they originate. EPA is building a database to track this information to consider key characteristics of individual PFAS to help guide forthcoming guidance on treatments.
In the meantime, Iyer advises operators to pay close attention to evolving developments and communications from EPA.
We recently saw the memorandum from EPA on addressing PFAS discharges in EPA-issued NPDES permits. We will look for guidance to the state permitting authorities to address PFAS in NPDES permits soon and more information from the EPA’s roadmap.
At SCS, we use our time to learn about technologies, including what’s still under investigation and explore what seems to work. In addition, watch for guidance documents, not just from EPA but from research organizations such as EREF and universities. Do your due diligence and keep your eyes and ears open for EPA and your state regulatory authority announcements. Staying informed is the best strategy for landfill operators at this point.
Liquids and wastewater management resources.
Not long ago, a Utah food manufacturer turned to SCS with a persistent problem: high concentrations of fats, oils, and grease (FOG) in its wastewater— high enough to clog the city’s sewer line, knock it out of compliance, and cost it a steep surcharge year after year. As the plant worked toward a solution, its customers’ demand was growing; it reached a point where it had to expand to keep up, and that’s when the quandary came to a head. The meat processor couldn’t get a permit for expansion until the FOG was in check.
Within 18 months, SCS Project Director Mark Pearson and his team of liquid management gurus had their client within acceptable discharge limits for the first time in years. Actually, the plant’s doing a lot better than meeting the city’s requirements. Its FOG concentrations, which had spiked to thousands of mg/L, are consistently down below the established discharge limit of 200 mg/L.
The scenario Pearson walked into is that the wastewater generation and pollutant loading were highly variable as flows fluctuated. Due to hydraulic limitations, the treatment system couldn’t keep up with volumes during peak flows. As a result, the influent (untreated wastewater) was discharged from the plant to the sewer to the municipal wastewater treatment plant. And because the system was overtaxed, it did not sufficiently break down the FOG, which exacerbated the problem, wreaking havoc with the city’s collection pipes.
After completing the initial assessment, Pearson’s team developed a multifaceted approach to debottleneck the system’s hydraulics and make other improvements to increase FOG removal efficiency.
Pearson; Dean Free, senior project manager; and Nathan Hamm, program lead for wastewater and liquids management, came up with a design that achieves two main goals: It eliminates uncontrolled discharge from the plant; it greatly reduces concentrations of FOG—cutting the contaminant load to the city. Not only is the client within discharge limits, but it’s also pushed through its most immediate barrier to expansion permit approval. And it’s improved its relationship with the city.
The solution is a complex one involving chemistry, mechanical engineering, and electrical engineering. But to pare a lot of fine details down to the nitty-gritty, Pearson says:
“We put in screens that remove solids in the wastewater. We constructed a 60,000-gallon tank to equalize the flow coming to the plant. We adjusted pH to help optimize the wastewater treatment chemicals’ effectiveness. We separated the influent tank from the effluent (treated water) pipes to solve a problem where the influent would overflow into the effluent through a perforated wall. The new piping setup makes overflow impossible. And to further fortify the system, we installed a lift station to capture previously untreated wastewater.”
As a bonus, the team installed a tank that the separated FOG residual is pumped to, then hauled to a compost facility for beneficial use.
Besides adding these system enhancements, the SCS team took capabilities further with a process control and instrumentation component. The technology monitors flow rates, temperatures, tank levels, and other conditions. It processes the data and automatically makes adjustments to achieve treatment goals, avoid production downtime, and enable operators to respond proactively should they see a red flag.
Comparing the old to the new way of monitoring and analyzing, Pearson says, “What our client had before was rudimentary information. And while they could monitor conditions, they had to walk around the plant. All of the operational data can be viewed on a screen now from one location. They have more data at their fingertips and more capability to make adjustments to avoid discharge exceedances.”
The wastewater treatment system upgrade was done as a design-build to speed the timeframe while also increasing efficiency. “We could immediately start rather than put it out to bid. We could do construction as we designed. And there was one entity and one point of responsibility. So what’s cool is we leverage both SCS engineering and construction capabilities to solve problems,” Hamm says.
He and his colleagues have the know-how to pivot on a dime if they have to, and there were a couple of times it was necessary, including when the client brought new management on board midway through the installation process. The new team preferred different instrumentation and had a specific scheme in mind.
“We were in the process of installing the original instruments. But we were able to incorporate their equipment preference midstream. We had to figure out how to get new instruments installed and ensure they were perfectly integrated with the computer control system that takes readings from the instruments. It was what they wanted, so we saw that they got it,” Pearson says.
While he and his co-workers’ jobs as project design and build engineers are done, they have not faded from the picture. They provide ongoing technical support when the client needs assistance with troubleshooting. The automated control system has helped.
“This is a robust and complex mechanical treatment system. If by chance, something was wrong, our client can transmit data that comes out of the process controller so we can work remotely to determine if process changes are needed. If they are, we can often make those changes from offsite, and quickly,” Hamm says.
But the SCS team also plans so that its client is equipped to ensure its success moving forward. They provide operator training. And they developed a standard operating and maintenance procedures manual and a checklist to track data and activities transferred from shift to shift, providing operators a standard and seamless way to communicate.
The busy Utah plant is on a good trajectory, with solid footing.
Says Pearson: “Before, they could not expand the plant or even continue their operations much longer if they did not get the FOG under control. Now they can operate continuously, discharge to the city, and they have potential to expand their plant because they are showing the regulators they can stay within their permit limits.”
Additional Resources and Case Studies:
Managing landfill leachate and wastewater treatment are increasingly challenging and costly for landfill owners and operators. In some cases, publicly owned treatment works (POTWs) are required to impose limitations on liquids received at their facilities, resulting in increased charges, or the POTW could refuse to permit or process the leachate wastewater altogether. These developments are due in part to more stringent discharge requirements and the shift to newer disinfection technology that has limited the POTW’s ability to accept higher strength wastewaters. As a result, many facilities and landfill operators are facing higher costs and fewer options for disposal.
Another factor that affects landfills is the fact that the composition of leachate in landfills differs depending on the degree of leachate stabilization and a seasonal increase in quantity as well as on the influence of more frequent and higher intensity storms due to changing climatic conditions.
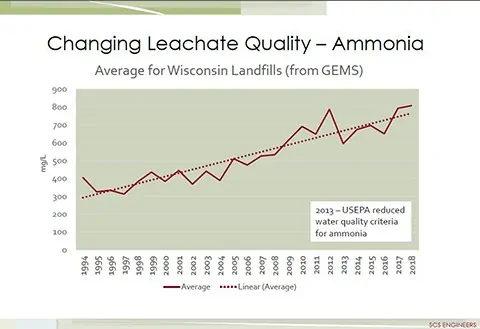
The single most influential factor on the volume of landfill leachate is precipitation. The most influential factor regarding leachate quality is that leachate typically contains high concentrations of organic compounds, ammonia and other forms of nitrogen, metals, and dissolved solids. Ammonia concentrations in the leachate, from many landfills, are increasing every year as shown in the graph below. Compounding the high strength concentrations of constituents found in landfill leachate are the emerging contaminants of concern including per and poly-fluoroalkyl substances (PFAS) that are now a significant concern with the U.S. EPA and many state environmental agencies.
Top 5 Questions and Answers When Selecting the Right Leachate Treatment Option for Your Landfill
Without considering leachate recirculation and a host of other factors, this blog provides answers to frequently asked questions regarding the analysis of treatment options for landfill leachate.
ONE: What is an example of a typical work scope of a leachate treatment options analysis?
TWO: What information is necessary to begin assessing the on-site treatment options of leachate?
THREE: What constituents should I expect to have analyzed to assess the options for leachate wastewater treatment?
FOUR: What are some of the issues taken into account regarding treating ammonia-N in leachate/wastewater?
FIVE: What are some examples of the options for how to effectively treat ammonia-N in leachate on-site?
We hope you find our SCS Advice from the Field blogs helpful. For more information, we recommend these articles and resources:
In this blog, we discuss the basics of various wastewater treatment methods in use today including process design parameters, advantages, disadvantages, and costs. Some of the wastewater treatment methods are better suited to landfill leachate treatment than others. For example, the traditional activated sludge wastewater treatment technology, discussed in this article is not suitable for landfill leachates, but it has been modified and improved so that its close cousin – for example the membrane bioreactor (MBR) – is much better suited for landfill leachate treatment. The blog is intended for those who manage projects that include leachate treatment but who are not wastewater or process engineers. We start by grouping treatment systems into three basic categories; biological, physical and chemical. Our first blog in the series focuses on biological treatment.
The oldest treatment technology for sanitary wastewater, with the longest track record, is biological treatment. It is effective for treating the type of wastewater generated by humans because it uses naturally occurring microbes to reduce organics, ammonia and other naturally occurring impurities. The modern version of this time-honored biological treatment process (from the 1960s forward) is known as the basic activated sludge (BAS) process. This is the type of treatment that is used at large publicly owned treatment works (POTWs) and the same form of treatment can be used, on a smaller scale, for landfill leachate. The wastewater to be treated, whether it is sanitary wastewater or landfill leachate, contains organic compounds, nutrients (e.g., nitrogen and phosphorus) and naturally occurring microorganisms. When given the right balance of soluble food, temperature, and oxygen, the microorganism population can be increased rapidly. In multiplying their numbers, the microorganisms absorb some of the food sources (organics) in the wastewater for energy and convert that to new cell mass (through cell growth and reproduction), carbon dioxide (CO2), and water.
The actual BAS treatment process involves pumping the raw wastewater through an open tank, aerating and agitating the liquid by mechanically adding air or oxygen typically through a set of fine bubble diffusers, then after a prescribed time, passing that water to another tank called a clarifier. The process is illustrated below.
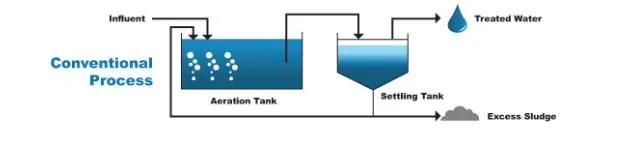
The dispersion pattern and the buoyancy of the bubbles promote mixing of the tank contents to ensure the bugs are getting adequate oxygen. An example of a bubble diffuser disc and a typical bubble diffuser bank layout is shown in the two photos below.

A process called nitrification also occurs in the aeration tank. Nitrification is a microbial process by which reduced nitrogen compounds (primarily ammonia) are sequentially oxidized to nitrite and nitrate by the species of microbes called Nitrosomonas, Nitrosospira, Nitrosococcus, and Nitrosolobus.
A biological system, such as a BAS has a susceptibility to some chemicals in leachate that in certain concentrations can be toxic to the “bugs.” For example, high concentrations of ammonia (NH3), chlorides or toxic substances can be harmful. Also, the treatment effectiveness of biological systems drops off significantly at a wastewater temperature lower than about 50 degrees Fahrenheit. Rapid changes in concentrations of these chemicals (or spikes) also can be harmful.
In the clarifier, flocculation chemicals are added to the water to aggregate and help to settle out the cell mass. The cell mass, almost 99% water, is called sludge. Periodically some of the sludge from the clarifier is removed and brought to the front of the process where this “activated” sludge (i.e., air enriched and microbes are alive) is used to seed the incoming wastewater with robust microbial growth and boost the growth of existing organisms that break the organics down. The balance of sludge (called “biosolid”) is removed from the clarifier and is typically dried, disinfected through use of high pH material and or heat and is then used as a soil conditioner directly or as a fertilizer ingredient.
The laboratory measurement of the amount of oxygen that microorganisms use to convert the food to new cell mass is called the biochemical oxygen demand (BOD5). The five day long BOD test was the earliest measure of the organic strength of wastewater and is a rough indication of how much energy (and relative cost) will be expended to treat the wastewater. Landfill leachate is typically considered a strong wastewater compared to municipal wastewater which is considered weak to moderate strength. Many municipal wastewaters are weak because they are heavily diluted with groundwater and infiltration of rainfall.
A key design parameter for any BAS process is the Mixed Liquor Suspended Solids (MLSS). The MLSS is the concentration of suspended solids in the aeration tank. The suspended solids are mostly the active microorganisms that do the work of decomposing the organic substances. The MLSS for a BAS is typically 4,000 to 6,000 mg/l.
Many other water chemistry aspects must be considered with the BAS process. However, these have not been included to simplify this explanation.
Over the decades the BAS process has been modified to address the many different types and strengths of wastewater, increase energy efficiency, reduce treatment times, improve resilience to shock loads, and pollutant removal effectiveness. You may have heard them called Sequencing Batch Reactor (SBR), or powdered activated carbon treatment (PACT). These are variations of the BAS process. Because landfill leachate is somewhat similar in chemical makeup to municipal wastewater, with typically higher chemical constituent concentrations, the landfill sector has successfully borrowed municipal treatment technology. An improvement to the BAS process that started showing up at landfills about 15 to 20 years ago is known as the Membrane Bio-Reactor (MBR) shown here.

The MBR took advantage of advances in micro-manufacturing capability in perfecting synthetic membrane filtration fabric. The membrane, when incorporated in the treatment process, eliminates the need for a clarifier. The membrane works by separating insoluble solids from the water. The advantages of the membrane filtration include;
The membranes are manufactured either as a cylinder shown below-left, or as a flat plate used in a rack outside the BAS reactor, or immersed in the BAS reactor. The diagram shown on the right illustrates the membrane filter process.

Some disadvantages of the MBR can include:
The MBR can manage a much higher MLSS than a conventional BAS; a typical maximum for a BAS is about 4,000 mg/l as compared to around 16,000 mg/l or more in an MBR. The higher MLSS allows for treating a stronger waste stream, and the system has better cold weather and shock load resistance. It can do this because the solids eventually are removed by a membrane. In the BAS process, the chemical treatment in the clarifier forms a large volume of sludge (organic chemicals + microorganisms) that has to be kept in balance by frequent sludge wasting out of the system. Without this wasting, the sludge volume recycled to the aerobic tank would overwhelm the aeration capacity, and the process would collapse. A process flow diagram may look like the one below.
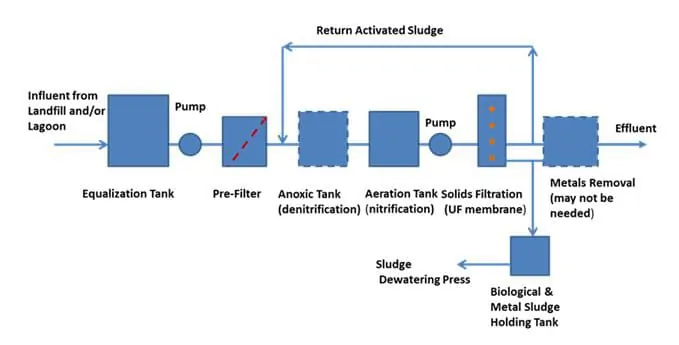
Typically, landfill leachate may need other treatment processes to supplement an MBR system to meet requirements for reducing specific pollutants and other parameters before the treated effluent can be released to the receiving water. One of the key parameters is ammonia (NH3) nitrogen.
Ammonia is produced in the landfill by microorganisms utilizing the organic substances in waste for energy and reproduction in an anaerobic (without oxygen) environment. In an aerobic environment outside the landfill, ammonia can be oxidized by bacteria in a process called nitrification, converting ammonia to nitrite and then to nitrate. When combined with enough phosphorus, nitrates released to surface waters in high enough concentrations can promote algae blooms. The algae blooms typically die off from cold, or their normal lifespan. Large amounts of dead algae compete for oxygen with fish and other wildlife. The result can be fish kills, which can further worsen the water body oxygen depletion problem.
So, in addition to the ammonia being converted to nitrite and then nitrate, the nitrate-nitrogen typically must be removed before discharge of the treated wastewater effluent to surface water. The nitrite and nitrate removal process is known as denitrification and involves reducing the nitrate to nitrogen gas which is inert and not harmful to the environment. Denitrification in the leachate treatment process can be accomplished either by ion exchange, chemical reduction, or biological processes.
The biological method is very common and is typically conducted in a tank known as the anoxic tank. The anoxic tank typically has a filter bed containing the nitrate conversion microorganisms where the nitrate-laden water is passed through for treating. No air is added to maintain an environment that is suitable for the heterotrophic microorganisms that convert nitrate to nitrogen gas. Methanol or acetic acid is typically added to provide a food source for the denitrifying culture in the filters.
Some other key process parameters for the MBR that will affect process sizing and footprint includes the following:
For example, using these unit costs an MBR plant rated at 65,000 GPD would have a basic capital cost of $1.2 to $1.4 million. This does not include a building. The annual operating cost of chemicals and energy only (not including labor), with 90% availability would be $640K to $1.07 million. Extra costs would include; a building if desired or necessary, electrical supply, equalization tank, yard piping, bench and pilot tests, and engineering. These are rough figures to give the reader a sense of the magnitude of costs. Actual costs will differ. It is always best to check with one or more vendors on the unit costs they experience with installations that they provide.
Biological processes have been adapted successfully from the municipal wastewater industry by the wastewater equipment manufacturers who service the solid waste industry to treat landfill leachate. They form the basis of a viable treatment platform that can be expanded depending on the effluent requirements and other design goals. By coupling high-quality membrane filters to remove solids with the BAS-type treatment modifications, MBR plants are revolutionizing leachate treatment. MBR plants are a more efficient and more effective treatment of higher strength leachates. Many vendors are offering modular systems that fit well with the leachate production and growth typical at landfills.
In our next blog, we will discuss membrane filtration systems that are increasingly finding application where leachate has to meet tough discharge standards. Follow SCS Engineers on Twitter, LinkedIn, or Facebook.
Learn more about Liquids Management – Leachate Services